Distribution of a novel serine synthetic enzyme — Proposal of serine synthetic enzyme identifiers
May 30, 2013
Enlarge this image
Whole structure of the novel PSP © Yoko Chiba.
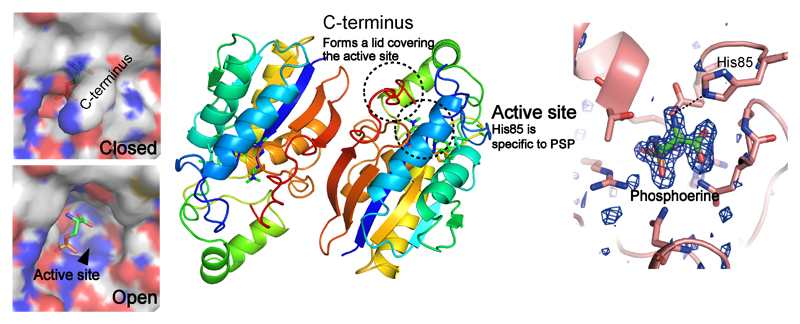
Todai researchers at the Graduate School of Agricultural and Life Sciences have discovered a novel-type phosphoserine phosphatase (PSP) from an obligate chemolithoautotrophic and hydrogen-oxidizing bacterium, Hydrogenobacter thermophilus. Homologs of the PSP exist in diverse organisms and it has been suggested that they are involved in serine biosynthesis. However, it has been very difficult to confirm the homologs as "real" PSP because of the diverse substrate preferences of each protein. Here, the researchers determined the crystal structure of the PSP to determine the structural units important for PSP activity.
The high resolution structure of the novel PSP revealed that His85, which can interact with its substrate, phosphoserine, and several carboxyl-terminal residues which can form a lid that covers the active site cleft were characteristic to PSP. Biochemical analysis using mutant proteins confirmed the importance of the two structural units for the novel PSP. Detection of high PSP activity only from novel PSP homologs from other organisms which conserved the two structural units confirmed them as important factors for predicting a protein working as a novel-type PSP.
These findings enabled the researchers to predict protein functioning as a novel-type PSP from genomic databases with high accuracy. As a result, not only the chemolithoautotrophic bacterium but diverse organisms such as cyanobacteria were found to have novel PSP with which they were synthesizing serine.
Paper Information
Yoko Chiba, Shoichiro Horita, Jun Ohtsuka, Hiroyuki Arai, Koji Nagata, Yasuo Igarashi, Masaru Tanokura, Masaharu Ishii, "Structural Units Important for Activity of a Novel-type Phosphoserine Phosphatase from Hydrogenobacter thermophilus TK-6 Revealed by Crystal Structure Analysis," The Journal of Biological Chemistry 288 (2013):11448-11458, doi: 10.1074/jbc.M112.449561.
http://www.jbc.org/content/288/16/11448

